Leaks After LAA Occlusion Tied to Uptick in Thromboembolic Events
In the past, the clinical impact of these leaks was uncertain, “but now it’s very clear that they matter,” Vivek Reddy says.
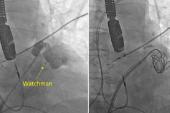
WASHINGTON, DC—When left atrial appendage occlusion (LAAO) was performed using the first-generation Watchman device (Boston Scientific), residual flow from the appendage was observed in about a quarter of patients at around 45 days, with the finding associated with a small but significant increase in thromboembolic events, according to a large registry analysis.
The rate of stroke, TIA, or systemic embolism was low overall, though it was significantly higher among patients with leaks of 5 mm of less—which accounted for nearly leaks—than among patients with no leaks (3.5% vs 3.0%; HR 1.15; 95% CI 1.03-1.29), Mohamad Alkhouli, MD (Mayo Clinic, Rochester, MN), reported at the American College of Cardiology (ACC) 2022 Scientific Session.
Only 0.7% of patients had larger leaks, and these were not associated with greater risks of adverse outcomes, likely related to the small size of the group and the fact that these patients were more likely to continue oral anticoagulation compared with those with smaller or no leaks, Alkhouli said.
This study, published simultaneously online in JACC: Clinical Electrophysiology, is the largest conducted so far looking at the clinical significance of peridevice leaks after LAAO, he noted. The vast majority of leaks were 5 mm or smaller, “which are considered not clinically significant as of today’s practice,” Alkhouli added. “Yet our study found that those small leaks were associated with 10% to 15% relative increase in risk-adjusted rates of thromboembolic and bleeding complications.”
‘Closure’ a Misnomer?
Alkhouli introduced his talk by questioning the name of the procedure itself. “Do we actually really close the appendage adequately or completely with this procedure, or is that a misnomer?” he said, pointing to rates of residual flow at 45 days ranging from about 10% to 50% across various studies of LAAO using different devices and imaging modalities. The frequency of the finding declines over time, but remains “not infrequent,” Alkhouli said.
He noted, too, that there’s no consensus regarding what defines a large leak, with some studies using a threshold of 3 mm and others 5 mm. “But we know that the average diameter of the middle cerebral artery is 3 mm, so that is a side note for you to decide what is important,” he said.
Whether leaks have a significant impact on clinical outcomes is not clear either. There was no relationship between residual leaks and thrombotic events in an analysis of PROTECT AF, with the first-generation Watchman device, and a study of LAAO using the Amplatzer Cardiac Plug (Abbott), but event rates were low. On the other hand, a more-recent registry study demonstrated worse clinical outcomes in patients with leaks larger than 3 mm. And a large study presented last year at the American Heart Association (AHA) 2021 Scientific Sessions by Vivek Reddy, MD (Icahn School of Medicine at Mount Sinai, New York, NY), showed that leaks smaller than 5 mm were associated with a higher rate of ischemic stroke/systemic embolism when they were still present a year after the procedure.
Our study found that those small leaks were associated with 10% to 15% relative increase in risk-adjusted rates of thromboembolic and bleeding complications. Mohamad Alkhouli
Alkhouli and colleagues explored the issue in a much-larger cohort of patients, turning to the National Cardiovascular Data Registry (NCDR) LAAO Registry. The analysis included 51,333 patients who underwent LAAO with the first-generation Watchman device between 2016 and 2019 and had a least a year of follow-up.
Most patients (73.4%) didn’t have peridevice leaks detected on echocardiography at around 45 days after the procedure; 25.8% had a small leak (≤ 5 mm) and 0.7% had a large leak (> 5 mm). Use of post-LAAO anticoagulation was similar in the patients with small or no leaks, with greater use seen among those with large leaks through 1 year.
There was a greater risk of stroke, TIA, or systemic embolism (HR 1.15; 95% CI 1.02-1.29) among patients with leak versus those with no residual flow around the device. Additional analyses revealed that leaks of 0 to 3 mm and 3 to 5 mm both appeared to be relevant when it came to risk of thromboembolic events, Alkhouli reported.
Similar to the analysis of thromboembolic events, a greater risk of major bleeding was seen when comparing small to no leaks (HR 1.11; 95% CI 1.03-1.20), despite the fact that anticoagulant use was similar in the two groups. Small leaks also correlated with a higher risk of major adverse events (death, cardiac arrest, stroke, TIA, systemic embolism, major bleeding, major vascular complication, MI, pericardial effusion requiring intervention, or device embolization).
Large leaks were not associated with any of the adverse outcomes. Residual flow, regardless of size, was not significantly associated with mortality, although deaths were numerically more frequent among patients with leaks larger than 5 mm. That could be related to the larger appendage diameter and higher rates of permanent atrial fibrillation and cardiomyopathy—indicating a sicker patient population—among those with large leaks, Alkhouli speculated.
Clear Results
Alkhouli acknowledged that the study was limited in that it used data from an observational registry that reflects variation in how leaks are measured and reported. Moreover, only the first-generation Watchman device was used during the study period, there was no information available on how leaks were managed, and follow-up was limited to 1 year.
Speaking with TCTMD, Reddy indicated that despite those issues, the analysis bolsters the one he presented at AHA 2021, which also showed an excess risk of thromboembolic events of about 0.5% per year in the presence of a leak. That analysis didn’t reveal a significant association between leaks present at 45 days and clinical outcomes—as seen in the LAAO Registry study—but that was likely due to insufficient patient numbers, an issue addressed by the current, much-larger analysis, he said.
“Leaks clearly matter,” said Reddy, who was the discussant after Alkhouli’s presentation. “In the past, we didn’t know whether or not they did based on some of the initial analyses with few patients, but now it’s very clear that they matter.”
Leaks clearly matter. Vivek Reddy
But the risk conferred by residual flow around the LAAO devices is “not terribly high,” Reddy said, who noted that many leaks present at 45 days after the procedure will close on their own over time.
Nevertheless, the findings underscore the importance of making sure the procedure is performed in such a way as to optimize closure of the appendage and minimize leaks, Reddy said. He added that use of newer devices, like the Watchman FLX (Boston Scientific) and Amplatzer Amulet (Abbott), and deflectable sheaths designed to facilitate implantation with the proper orientation—which both reduce leaks—will change considerations around the issue.
But the main concern at this point is what to do about residual flow when it’s there, Reddy said. “Early leaks do seem to increase the risk. The magnitude of the risk is not that great, so I think it’s probably okay to just wait to see if they close, and if they close, then you’re okay,” he said. “If they don’t close, then we probably have to think about whether or not to close them.” Options include methods like embolization coils and plugs, although solid data supporting those approaches are lacking.
Alkhouli said the data provide a mixed picture, with both reassuring and concerning results. On the positive side, the absolute risk tied to leaks is small. “Yet it is a preventative procedure, so we would like to get it as good as we can,” he added.
The results should not be used to close small leaks in all patients using coiling, for instance, because there are limited data indicating that the approach works over the long term, Alkhouli said.
“I think we should study this more with the current practice, with the newer devices, with the current techniques,” he said, making a pitch for a randomized trial—perhaps with longer-term anticoagulation as one of the comparators—to provide some answers.
In the meantime, Alkhouli concluded, “it’s good to have newer technologies that are trying to get as much of an occlusion as we can.”
Todd Neale is the Associate News Editor for TCTMD and a Senior Medical Journalist. He got his start in journalism at …
Read Full BioSources
Alkhouli M, Du C, Killu A, et al. Clinical impact of residual leaks following left atrial appendage occlusion: insights from the NCDR LAAO registry. J Am Coll Cardiol EP. 2022;Epub ahead of print.
Disclosures
- The study was funded by a grant from Boston Scientific.
- Alkhouli reports serving on the advisory board of Boston Scientific.
- Reddy reports consultant fees/honoraria from Abbott, AFTx, AtriAN, Biosense Webster, Biotronik, Boston Scientific, Cardionomic, CardioNXT, CoreMap, EBR, Fire1, Impulse Dynamics, Medtronic‐Cryocath, and Philips; stock options from Acutus Medical, Affera, Apama Medical, Autonomix, Axon, BackBeat, BioSig, Circa, Corvia Medical, East End Medical, EP Dynamics, Epi EP, Epix Therapeutics, Eximo, Farapulse, Javelin, Keystone, LuxCath, Medlumics, Middlepeak, NewPace, ValCare, and VytronUS; ownership interest/partnership/principal in Manual Surgical Sciences; and stock from APN Health, Biostar Ventures, CoRISMA, Dinova Hangzhou Nuomao Medtech, HRT, Intershunt, Kardium, Newtrace, Restore Medical, and Surecor.




Comments