RAPID CABG: No Penalty to Early Surgery for ACS After Stopping Ticagrelor
Quicker CABG can lessen the “major headache” of patients waiting and degrading in-hospital, says Christopher Cannon.
(UPDATED) In patients with ACS, performing CABG within a couple of days after ticagrelor cessation is noninferior to waiting 5 or 6 days in terms of bleeding events, according to new randomized data. Moreover, patients with delayed CABG had more presurgical ischemic events and longer hospital stays.
 To prevent bleeding, current US guidelines recommend waiting at least 5 days to perform nonurgent CABG on patients with ACS who had been taking ticagrelor, while European guidelines shorten that period to at least 3 days.
To prevent bleeding, current US guidelines recommend waiting at least 5 days to perform nonurgent CABG on patients with ACS who had been taking ticagrelor, while European guidelines shorten that period to at least 3 days.
“All of the guidelines are based on cohort studies and pharmacodynamic studies with no randomized evidence,” said Derek So, MD (University of Ottawa Heart Institute, Canada), who presented the RAPID CABG findings today at the virtual American Heart Association (AHA) 2021 Scientific Sessions. This trial “is the first and only randomized study to evaluate the safety of early surgery,” he added.
The half-life of the P2Y12 inhibitor is 12 hours, according to Christopher P. Cannon, MD (Brigham and Women's Hospital, Boston, MA), so “waiting 3 days is more than four half-lifes.” Commenting to TCTMD that RAPID CABG “might be my new favorite study,” he said the findings are “going to be a major help for surgeons and cardiologists managing patients with acute coronary syndrome who need CABG.”
[This] might be my new favorite study. Christopher P. Cannon
“This has been an issue for two decades and it’s a major headache, especially now that our hospitals are so full, to have people just sitting around waiting and doing nothing and then at risk, as they show, of having recurrent angina heating up again,” Cannon continued, noting that some doctors are already performing CABG sooner than the guidelines recommend after stopping therapy.
Commenting during a press briefing, Joanna Chikwe, MD (Cedars-Sinai Medical Center, Los Angeles, CA), said she is one of those surgeons. “You can ask any of my partners: I'm comfortable going within 48 hours,” she said. “I'm really glad to see this data. It confirms what I think is safe.”
To TCTMD, Michael Borger, MD, PhD (Leipzig Heart Center, Germany), said the results of RAPID CABG will “definitely” change his surgical practice. “This trial clearly shows that 2-3 days is not only safe but probably even effective in that it seems to also reduce the ischemic events that can occur during the waiting period,” he said. “Before, we were basically going with 5 days, but now I will be quite confident with shortening that to 2-3 days like the investigators demonstrated.”
His confidence is bolstered by the “pretty convincing” P value for noninferiority “even though the sample size was not that large,” Borger added.
Todd Rosengart, MD (Baylor College of Medicine, Houston, TX), said he, too, will change his practice based on the findings. “We've been long proponents of a more-tailored withhold of antiplatelet agents,” he said, noting that “the next step is not an arbitrary time limit” but platelet testing. “There's about a 30-30-30 split genetically in terms of platelet reactivity. So one size does not fit all, and we'd actually recommend a platelet aggregometry testing.”
Additionally, Shemy Carasso, MD (Poriya Medical Center, Tiberias, Israel), said the findings are in line with a recent similar study, by his own research group, that also did not show an increase in complications when ticagrelor was stopped only the day before surgery.. “These data are reassuring [and] probably practice changing, suggesting that further delay in urgent CABG to avoid the drug effect may not be necessary,” he told TCTMD in an email. “Surgeons just need to know and be ready.”
Similar Bleeding, Fewer Ischemic Events
For the physician-initiated multicenter study, So and colleagues randomized 143 patients with ACS (mean age 64 years; 93% multivessel disease) slated for surgery to undergo CABG within 2-3 days or 5-7 days after stopping ticagrelor. In the early group, six patients were operated on later than the assigned window due to logistic/medical reasons, and in the delayed group seven patients underwent CABG sooner than allocated for medical/physician reasons and four went later due to logistic/medical reasons.
Median time to CABG was 3 and 6 days in the early and delayed arms, respectively. Also, prebypass platelet reactivity by VerifyNow was significantly lower in the former group (200 vs 251 PRU; P < 0.001).
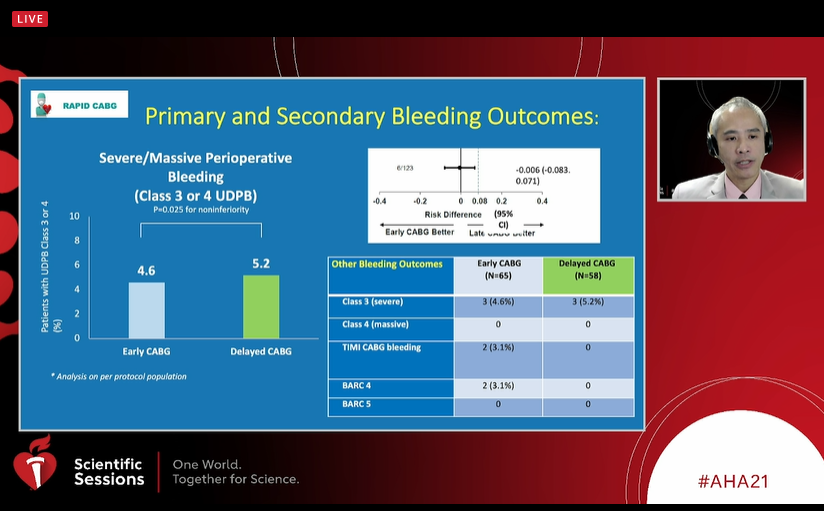
The primary outcome of severe/massive bleeding, as defined by the Universal Definition of Perioperative Bleeding class 3 or 4, was reported in a similar proportion of patients in both the early and delayed CABG cohorts, with all bleeds being classified as severe (4.6% vs 5.2%; P = 0.0253 for noninferiority with an 8% margin). Also, two instances of TIMI CABG and BARC 4 bleeding were seen in the early CABG arm, whereas none were documented in the late CABG group.
However, while no ischemic events were identified in those randomized to early CABG, the rate of presurgical events among patients waiting for later CABG was 8.7%, including one MI, four cases of recurrent angina, and one ventricular tachycardia. Median hospital length of stay was also shorter for patients who received CABG sooner rather than later (9 vs 12 days). At 6 months, the cumulative MACE rate was lower with early versus delayed CABG (5.6% vs 13.0%).
“Although our study was not large enough to evaluate whether early surgery protects people from suffering early recurrent heart attacks or angina, it opens this hypothesis and may prompt future research in the area,” So said in a press release.
Speaking during the press conference, AHA program committee chair Manesh Patel, MD (Duke University School of Medicine, Durham, NC), said while the noninferiority margin was wide in RAPID CABG, “the actual observed difference was . . . reassuring.”
I'm really glad to see this data. It confirms what I think is safe. Joanna Chikwe
Cannon added that the “modest” size of the study, especially for a surgical trial, along with the consistent endpoints, including the various bleeding scales included, are “encouraging.”
More generally, Chikwe commented that her “broader concerns about how we design and conduct trials in cardiac surgery . . . are really driven by inherent challenges. [It's] very hard to recruit patients” compared with a hypertension trial, for example.
In cardiac surgery trials, Chikwe continued, “it's almost a given that you will need to use surrogate endpoints, and the choice of the surrogate endpoints can determine which way the trial finds, whether it's in favor or against what you are looking at. So, I think the RAPID CABG trial is helpful—a small number of events in the aggressive early-intervention arm could have flipped the finding the other way. Which is why, although it confirms my practice, I think physicians may want to see a larger-powered trial to be convincingly compelled that they should change their practice.”
Yael L. Maxwell is Senior Medical Journalist for TCTMD and Section Editor of TCTMD's Fellows Forum. She served as the inaugural…
Read Full BioSources
So DYF. A randomized study of early vs. delayed coronary artery bypass surgery among patients with acute coronary syndromes treated with ticagrelor: the RAPID CABG study. Presented at: AHA 2021. November 13, 2021.
Disclosures
- RAPID CABG was sponsored by the Canadian Institute of Health Research and an ORACLE grant – University of Ottawa Heart Institute.
- So reports receiving grant/research support from Spartan Biosciences, Aggredyne, Roche Diagnostics, and Fujimori Kogyo; serving as a consultant for Bayer Canada, AstraZeneca Canada, and JAMP Canada; and serving on the speakers’ bureau for AstraZeneca Canada, Servier Canada, and HLS Therapeutics.
- Cannon reports receiving research grants from Amgen, Better Therapeutics, Boehringer-Ingelheim, Bristol-Myers Squibb, Daiichi Sankyo, Janssen, Merck, Novo Nordisk, and Pfizer; receiving consulting fees from Aegerion/Amryt, Alnylam, Amarin, Amgen, Applied Therapeutics, Ascendia, Boehringer-Ingelheim, Bristol-Myers Squibb, Eli Lilly, Janssen, Lexicon, Merck, Pfizer, Rhoshan, and Sanofi; and serving on Data and Safety Monitoring Boards for the Veteran’s Administration, Applied Therapeutics, and Novo Nordisk.
- Rosengart, Borger, and Carasso report no relevant conflicts of interest.




Comments